Holistic healthcare,
for
women
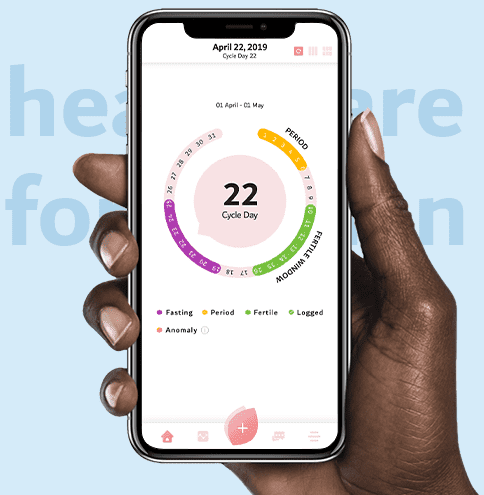
24/7 support for complex female health issues.
Subscription-based care accessible from work and home.
Designed to complement health insurance.
24/7 support for complex female health issues. Subscription-based care accessible from work and home. Designed to complement health insurance.