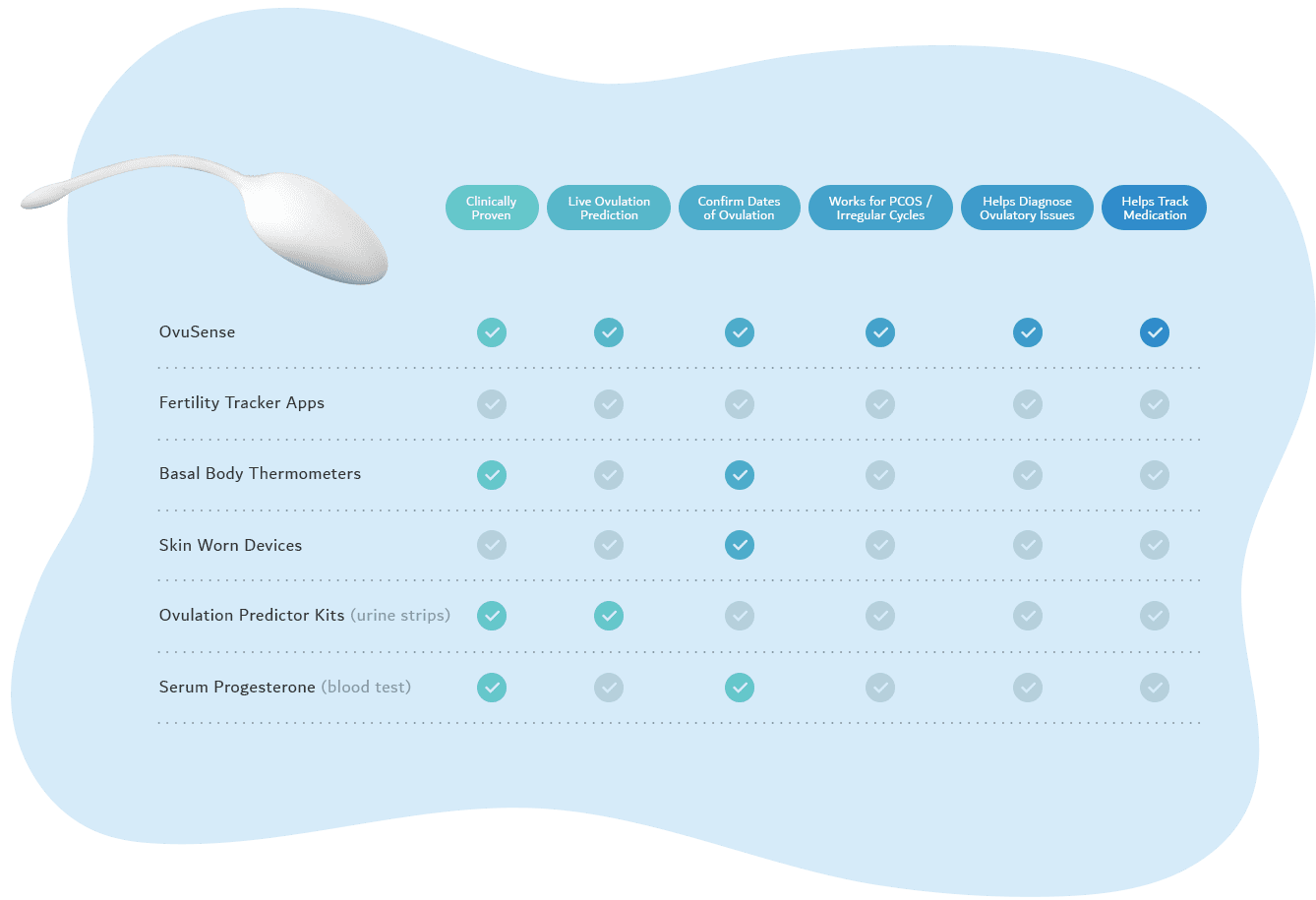
OvuSense
™ is an
FDA-approved, CE-certified medical device.
[a] The positive predictive value measures how many of the predicted ovulations OvuSense
™ gets right.
[b] The accuracy measures how many positive and absent ovulations OvuSense
™ confirms correctly.
[c] Note: 4 of the OvuSense
™ publications were clinical posters shown at the annual meetings of the two largest clinical societies for reproductive medicine: ESHRE and ASRM. Poster publications are reviewed by a committee of peers for both societies and only allowed to be presented if deemed of sufficient scientific merit and robustness. They are then published as part of the proceedings of the meetings in the official society journals ‘Human Reproduction’ and ‘Fertility and Sterility’, respectively. As is the norm with the development of medical devices, the OvuSense
™ trials were sponsored by the company but the primary authors of the posters are independent clinicians.
[1] The key publication outlining prediction of ovulation was shown at the 2014 ESHRE meeting:
Papaioannou S, Delkos D, Pardey J (2014).
[2] The claims for accuracy are based on the data set shown in [1], originally published at the 2013 ASRM meeting as a quality index:
Papaioannou S, Aslam M (2013).
[3] US Patents:
US8684944,
US8496597,
US9155522,
US9155523,
US20190110692; European patents:
EP1485690,
EP2061380; Chinese patent:
CN1650154.
[4] The comfort and ease of use of OvuSense
™ were published in the Journal of Obstetrics and Gynecology in 2013, and this survey is repeated annually with our customers with continuous as good or better results:
Papaioannou S, Aslam M (2013).